News
News items will be published here, but also sign up to our Newsletters — see the form on the right.
You can receive updates from the News feed, either to your RSS reader or via email.
We need your help - again!
In this issue:
- CNHC Campaign
- Reflexology advertising
- Hypnotherapy discipline descriptor update
- Marigold therapy update
 CNHC Campaign
CNHC Campaign
In our last newsletter, we asked supporters to respond to the Professional Standards Authority's call for information on the Complementary and Natural Healthcare Council's application to join their Accredited Voluntary Register.
We also asked our supporters to submit complaints to the CNHC to highlight that many of their registrants were making claims outside of their advertising guidance, etc.
This involved filling in a pdf and adding a scan of your signature, so we've decided to simplify the process: you can now send the details to us using our simple online form: CNHC Campaign.
It'll only take you a few minutes to find a CNHC registrant making claims that you think might be misleading and send us the details.
We will collate the responses, check and submit them to the CNHC.
So please do your bit to help us and find claims that concern you. Submit as many as you like and we'll take care of the rest.
Warning: once you've started, you may find it addictive! Get going now by filling in the form.
Reflexology advertising
You may remember that one of our first campaigns was about the claims made by reflexologists. We won the ASA 'master' complaints, of course, and the ASA have been working to persuade reflexologists to comply with the ASA's CAP Code and guidance.
Many reflexologists are registered with the CNHC and we were pleased to read their April 2013 newsletter:
CAP Compliance Team focuses on reflexology websites
Following the notice about reflexology advertising in our July 2012 news update, the Committee of Advertising Practice (CAP) Compliance team is now actively checking the websites of reflexologists to ensure the wording used meets the Advertising Codes. We strongly encourage all CNHC registered reflexologists to check CAP’s website here http://www.cap.org.uk/Advice-Training-on-the-rules/Advice-Online-Database/Therapies-Reflexology.aspx and to use the wording provided in CNHC’s reflexology descriptor which is available to download by logging into My CNHC or to view here: Complementary therapy descriptors.
Apart from saying what a reflexologist believes, all their therapy descriptor says about what they can claim is:
Reflexology can be a wonderfully relaxing experience where you can take time out from everyday pressures. The therapist’s expert touch will help you relax which can help improve mood, aid sleep and relieve tension. The result is an overall sense of wellbeing.
We're sure it can be relaxing, but there is no good evidence that it can do any more than that — and certainly none that would meet the ASA's requirements for substantiation. But while many are meeting their responsibilities, others are still making claims well outside their therapy descriptor and the ASA's guidance.
It's sadly all too easy to find reflexologists claiming:
Some of the conditions reflexology can help with are as follows:
Nervous system: headaches, migraines, insomnia, emotional stress, anxiety, panic attacks, depression.
Endocrine system with hormonal imbalances, menstrual problems, menopause, PMT, Sub fertility/infertility, prostate issues, thyroid imbalance.
Circulatory and respiratory systems with hypertension (high blood pressure), hypotension (low blood pressure), poor circulation, asthma, sinusitis, Hay fever, bronchitis, pneumonia.
This is why we need to bring what reflexologists — and other CNHC registrants — are claiming to the attention of the CNHC so that they realise the extent of the problem they have on their hands and take whatever steps are necessary to resolve it.
They are supposed to act in the public interest but cannot be doing that if they continue to allow misleading claims to be made by their registrants.
That is why our CNHC campaign is so important and we urge you to help us by doing what you can.
Hypnotherapy discipline descriptor update
After our ASA complaint about the claims being made by the Royal London Homeopathic Hospital in their Medical & Clinical Hypnosis leaflet, the ASA updated their guidance on hypnotherapy in line with the evidence and the CNHC have now done the same with their therapy descriptor. In the same newsletter, they say:
CNHC’s hypnotherapy discipline descriptor has been updated in consultation with the Hypnotherapy Profession Specific Board and the Committee of Advertising Practice (CAP) Copy Advice Team. The update means that CNHC registered hypnotherapists may now refer to the use of hypnotherapy for Irritable Bowel Syndrome (IBS) using the wording suggested. This follows discussions with CNHC Board member Professor George Lewith about the evidence for the use of hypnosis with IBS.
Sage and timely advice, but it doesn't really tell the whole story about the claims for IBS the ASA will now allow.
In their adjudication, the ASA said:
We noted the quality of the studies varied significantly, but each had shown that hypnotherapy/hypnosis had some effect on helping patients to deal with the pain or discomfort from their conditions. However, we considered that the studies had not supported the claim that medical hypnosis could treat or cure gastrointestinal disorders. We therefore concluded that the claim "The following medical problems have been shown to benefit from the use of medical hypnosis: Gastrointestinal Disorders - Irritable Bowel Syndrome - Chronic Functional Abdominal Pain - Gastro-oesophageal Reflux Disease - Functional Dyspepsia" was misleading.
This clearly states that they will allow claims about hypnosis helping with the pain or discomfort from their conditions, but not claims about the actual conditions themselves. This is an important point advertisers will need to take on board if they are to stay on the right side of the CAP Code.
Marigold therapy update
You may also remember our other complaint about claims made in another RLHIM leaflet was for their marigold therapy for various foot problems. After providing evidence to substantiate their claims, the RLHIM simply withdrew the leaflet and the case was informally resolved.
As part of our ongoing investigations into marigold therapy, we submitted complaints about three websites to the MHRA after they confirmed to us that the marigold product was not not registered and not authorised by the MHRA. As they were complaints about the advertising claims made, the MHRA passed them to the ASA to deal with.
They were all informally resolved by the ASA: the first (Integrated Medical Centre Ltd) was published on 3 April and the other two (The Marigold Trust and Marigold Foot Care Ltd) were published on 24 April. The page on the first website that mentioned marigold therapy has been removed and the other two websites have been completely taken down.
We have a lot more to tell you about this, but that's all we can say at the moment!
25 April 2013
CNHC - Time for Action
There is a regulator of 'complementary' practitioners that is supposed to be protecting the public. How well are they performing that duty?

Last July we were pleased to report on the sensible advice on advertising issued by the Complementary and Natural Healthcare Council (CNHC) to registrants after the ASA upheld our master complaints on reflexology.
This advice on top of their Guidance Sheet on advertising certainly gives the impression that the CNHC takes the issue of practitioners making "inappropriate or unsubstantiated claims" seriously and quite right too — especially given the CNHC has just applied for Accredited Voluntary Register status with the statutory Professional Standards Authority (PSA) — the new name of the CHRE.
When considering an application to become accredited, the PSA issues a 'Call for evidence':
Organisations applying for accreditation will have to demonstrate (by providing evidence) that they meet the standards set out by the Professional Standards Authority. The Authority will check, challenge and confirm the evidence provided by the applying organisation and will take account of feedback from patients, service users, the public, professional and representative organisations and others.
We would like to hear the experiences you have had with organisations applying for accreditation. Your contribution can be as lengthy or as brief as you like, but we will only be able to take it into account if it is supported by evidence rather than just being an expression of your opinion or based on hearsay (i.e. what someone else has told you).
We think the PSA need to be asking some probing questions of the CNHC and we will be submitting our own response. If you have information you think is relevant, we urge you to do so as well — but note the deadline is Wednesday, 3 April 2013.
To provide your feedback, please complete their form and email it to This email address is being protected from spambots. You need JavaScript enabled to view it..
History
But to help understand why we are concerned about the CNHC being granted AVR status, we need to briefly look at their history.
The birth and ongoing problems with the CNHC have been covered by Andy Lewis on The Quackometer and the problems they had with dealing with complaints about their registrants by Simon Perry:
The CNHC wishes to place on formal record their thanks to Simon Perry
In summary, as a result of Simon's complaints, the CNHC stated:
- CNHC will tell practitioners to remove claims they cannot justify.
- CNHC will conduct a review of evidence base for regulated therapies.
- CNHC will contact all registrants to instruct them not to make claims without justification.
- CNHC will contact complementary health course providers and authors to instruct them not to make claims without justification.
A responsible regulator would have already done all that, particularly one whose stated aim is to act in the public interest. But it's good they realise they had to do this to protect the public from being misled by their registrants.
Progress? What progress?
But what progress have they made?
The CNHC originally produced 'Therapy Descriptors' for each therapy they registered. These detailed what the therapist could do for you and what you could expect at a session. For example, their Therapy Descriptor for reflexology originaly stated:
How reflexology may help
Numerous disorders may benefit from reflexology depending on the specialism and expertise of your practitioner. Examples include pain, headaches, sinus problems, hormonal imbalances, back problems, stress and tension.
This clearly misleads the public into thinking that reflexology can help with those conditions. After lengthy discussions with the Advertising Standards Authority (ASA), these therapy descriptors had to be re-written and are now CAP Code friendly, with no mention of any medical condition anywhere. All they are left with are vague notions of 'well-being' and 'relaxation' and what a practitioner will do to you. Read them all here.
For example, the new reflexology descriptor now simply says:
Reflexology works on an individual basis and may alleviate and improve symptoms such as everyday stress and tension.
We have no doubt many of these therapies are relaxing, but there is little or no evidence they can do any more than that.
Any CNHC registrant keeping within these new descriptors is far less likely to be challenged and far less likely to mislead an unsuspecting public.
Making a difference
With these therapy descriptors in place for — in many cases — well over a year, have CNHC registrants removed misleading claims from their websites? Have the CNHC enforced their advertising rules (never mind what they promised Simon)?
No doubt some registrants, aware of their responsibilities and wanting to live up to them, will have changed their websites, but swathes of claims well outside the therapy descriptors remain across a wide range of therapies.
It is clear that, however well intentioned the CNHC may be, they are not being vigilant enough about what their registrants are claiming. If registrants are making claims that cannot be substantiated with good evidence, their customers cannot give their fully informed consent to treatment and that is a critical breach of their Code of Conduct, Performance and Ethics.
So, we need to make them aware of the extent of these claims and we want to see how they deal with registrants as a test of their professionalism and ability to properly protect the public.
We are therefore asking our supporters to submit complaints about questionable claims made by CNHC registrants.
Submitting a complaint
CNHC registrants can be found by choosing a particular therapy and searching for registrants in your area using their Search facility. But first, familiarise yourself with these CNHC documents:
- What types of complaints against registrants can we consider?
- Code of Conduct, Performance and Ethics
- Advertising Guidance
- Complaints Procedure
- Complaints Handling Process
Once you have found a registrant who you believe may be making claims about the therapy for which they are CNHC registered that you don't think can be properly substantiated or that go beyond the Therapy Descriptors or the ASA's CAP Code and guidance, you can submit a complaint. Please check carefully to ensure that it is a CNHC registrant who is making the claims and for a therapy for which they are registered.
Submitting the complaint can be done in several ways: using FishBarrel (which has recently been updated to include the CNHC's complaints form) or by filling the CNHC's complaints form manually. Because this form is a simple pdf, we have turned it into one that can be filled in electronically — download it here. Remember that you need to sign the form and that the CNHC are unlikely to accept any complaint not presented on their form.
Some suggested overall text can be downloaded here, but please feel free to use your own words or adapt the text to suit. But please remember these rules:
- Keep it simple and to the point;
- Keep it impersonal and factual;
- Make sure you give the name of the registrant, his/her therapy and the claims for that therapy you are questioning.
If you use our electronic version of the form, you should be able to fill it in electronically in your pdf reader and add a scan of your signature. To do this on recent versions of Adobe Reader, click on 'Sign' at the top right of the Reader, then 'Place signature', then choose 'Use an image' to add your pre-scanned signature into the form in the correct place. Make sure your text is fully visible on the form, but if it is longer than the text box will allow, add it as an additional sheet (Word, text, etc) and list it on the form under Additional Information. Remember to keep a copy of everything you send. Ideally, take a snapshot of all web pages you are complaining about and monitor them for changes.
The completed form can then either be sent by post to the address given on the form, or emailed to This email address is being protected from spambots. You need JavaScript enabled to view it. (FishBarrel will do this automatically) marked for the attention of the Fitness to Practice Officer.
Please tell us when you submit a complaint and when it has been resolved to your satisfaction.
27 March 2013
WDDTY #9 - Taking Stock
In this issue:
- What Doctors Don't Tell You — OxyTech
- What Doctors Don't Tell You — Taking Stock
- Holland and Barrett — The final complaint?
 OxyTech
OxyTech
This is Dulwich Health's second appearance here. It's another food supplement; this time one called OxyTech. The advert in October's issue of What Doctors Don't Tell You said:
For candida, bloated stomach, irritable bowel, leaky-gut, skin disorder, continuous constipation or diarrhoea and lack of oxygen and energy.
OxyTech regenerates your colon lining, so that it functions normally.
Can children take OxyTech? It is ideal for children at any age who are constipated or are taking antibiotics. If the child is too small to swallow capsules, the contents from a capsule can be spread on food, mixed in a smoothie, taken with honey etc.
If I get food poisoning? Take a large dose of OxyTech (say up to 10 capsules) as soon as possible.
My baby has colic? If you are breast feeding, you take OxyTech to regulate your body. If bottle feeding give baby OxyTech powder by teaspoon mixed with honey.
In their adjudication published today, the the last time ruled that because OxyTech was a food supplement, the CAP Code prohibited:
…marketing communications that claimed that a food could prevent, treat or cure human disease, and required reduction-of disease-risk claims and claims that referred to children’s development and health to be authorised by the European Commission. Because the ad made prohibited claims and we had not seen evidence that the reduction-of disease-risk claims and claims relating to children’s health were authorised by the European Commission, we concluded that it breached the Code.
Breaching the CAP Code on two counts, the ASA told Dulwich Health:
The ad must not appear again in its current form. We told Dulwich Health Ltd not to state or imply that their products could prevent, treat or cure human disease. We also told them not to make reduction-of disease-risk claims and claims that referred to children’s development and health unless authorised by the European Commission. We referred the matter to CAP's Compliance team.
In response to the ASA, Dulwich Health said:
Dulwich Health Ltd acknowledged receipt of our letter and said they noted our comments.
Just like the last time, they made no attempt to justify or substantiate the claims they made.
But this time, the ASA have referred the matter to their Compliance Team who will try to work with Dulwich Health to ensure future marketing complies with the CAP Code. We hope that Dulwich Health will cooperate and take their responsibilities under the CAP Code seriously to ensure all their marketing — including that on their website — complies.
It's worth emphasising that, in general, the same rules apply to all advertisers, regardless of the product or service they are marketing. There are, rightly, some additional rules for adverts about health products, but all advertisers have an obligation to ensure their adverts comply, whether they are advertising food supplements, double glazing, solar panels or car insurance.
Taking stock
Last October, we started to tell you about What Doctors Don't Tell You, and that we had submitted complaints about 26 adverts that had appeared in the first two issues. The following table shows our progress (the advertisers' names link to our news item about them and with the advertisers' website added):
| Advertiser | Website | Product/service | Issue/s | ASA outcome published |
| www.learnbuteyko.co.uk | www.learnbuteyko.co.uk | Buteyko breathing method | September | 14 November 2012 |
| Wholistic Research | www.wholisticresearch.com | Water filter | September October |
14 November 2012 |
| General Naturopathic Council | www.gncouncil.co.uk | Naturopathy, iridology | September | 12 December 2012 |
| General Naturopathic Council | www.gncouncil.co.uk | Naturopathy | October | 12 December 2012 |
| General Naturopathic Council | www.gncouncil.co.uk | Naturopathy | September | 12 December 2012 |
| General Naturopathic Council | www.gncouncil.co.uk | Naturopathy | October | 12 December 2012 |
| www.asthmacarekent.co.uk | www.asthmacarekent.co.uk | Buteyko breathing Method | October | 12 December 2012 |
| CoMra Therapy | www.comra-therapy.co.uk | CoMra device | September | 12 December 2012 |
| CoMra Therapy | www.comra-therapy.co.uk | CoMra device | October | 12 December 2012 |
| James White Drinks Ltd/Beet-it | www.beet-it.com | Beet-It Beetroot juice | October | 19 December 2012 |
| Medical Thermal Imaging Limited | www.breast-angels.com | Thermography service | September | 09 January 2013 |
| Abundance & Health Ltd | www.lyposphericnutrients.co.uk | Lypo-shperic supplement | September October |
09 January 2013 |
| Simply Vital | www.simplyvital.com | vitalCALM supplement | September | 09 January 2013 |
| Stetzer UK | www.stetzer.co.uk | Mains filter device | September | 16 January 2013 |
| Good Health Naturally | www.goodhealthnaturally.com | Serrapeptase supplement | September | 16 January 2013 |
| Clarus | www.qlinkeurope.eu | Q-link device | September October |
23 January 2013 |
| BEP Technology Ltd | www.bioenergyproducts.co.uk | Earthing device | September October |
06 February 2013 |
| Dulwich Health Ltd | www.dulwichhealth.com | AlliTech supplement | September | 06 February 2013 |
| Harmony United Ltd | www.harmonyunited.com | Health device | September | 13 February 2013 |
| Harmony United Ltd | www.harmonyunited.com | Health device | October | 13 February 2013 |
| International Antiaging Systems Ltd | www.melatoninznse.com | Melatonin Zn Se | September | 13 February 2013 |
| Dulwich Health Ltd | www.dulwichhealth.com | OxyTech supplement | October | 27 February 2013 |
Key: Informally resolved case, Adjudication
These outcomes gives us:

With 54 CAP Code breaches in just nine adjudications on eleven adverts and complaints on another eleven adverts informally resolved, we now have a further four still to be resolved.
But we've already had to submit a complaint about one advert to the medicines regulator, the MHRA, because the advertiser didn't seem to recognise the authority and competence of the ASA.
Several of the other issues were about medical claims made for supplements — will they also have to be reported to the MHRA if they continue to make similar claims in the magazine or on their websites?
Holland and Barrett
This will hopefully be the last time the high street chain gets a mention here.
Despite previous assurances that all the prohibited point-of-sale advertising material for the Nelsons homeopathic products they sell had been removed from all their stores last summer, one of our supporters told us this was not the case. Last October, we submitted a third complaint to the medicines regulator, the MHRA, with a photo of prohibited material in their store in the Trafford Centre in Manchester. The complaint was fairly quickly resolved, but it has taken the MHRA nearly four months just to publish the details on their website.
In their notice, published on Monday, the MHRA said:
This was the third complaint around this issue, which was previously investigated and upheld in November 2011 and July 2012. Holland and Barrett had previously agreed to withdraw the materials from all stores and implement measures to try to ensure this would not happen again.
The MHRA upheld this complaint and warned the company about compliance. Holland and Barrett immediately ensured the material was withdrawn from the store subject to this complaint. They issued another recall to all Holland and Barrett stores this time requesting that all stores confirmed no Nelsons materials were present and introduced more robust procedures to ensure future compliance.
Our original complaint was about the booklets that were part of the display stands (and we have also been sent photos of other advertising cards). These booklets contained indications linked to the homeopathic products. Under the terms of the registration of these products, indications for what they might be used for are prohibited. We also originally complained about the labels on the display stand that said: 'First Aid', 'Aches & Pains', 'Skin, 'Colds & Infections', Women’s health' and 'Digestion'. We believed these too could be seen as prohibited indications, so we reminded the MHRA that these had also been part of our complaint. The MHRA told us:
We had forwarded your concerns regarding the display labels and H&B were not able to gain justification from Nelsons for their use, hence the decision to remove them.
Holland and Barrett issued yet another memo to all their stores on 30 November 2012 to ensure all prohibited advertising had been removed:
You must remove all Nelsons literature including any flip books that are attached to stands immediately; otherwise we will be facing enforcement action from the MHRA/IMB.
They included this diagram to illustrate what should be removed:
 We sincerely hope this is the end of the matter and that no store — whether Holland and Barrett or otherwise — now misleads the public by displaying prohibited advertising material. But if you see any, please let us know.
We sincerely hope this is the end of the matter and that no store — whether Holland and Barrett or otherwise — now misleads the public by displaying prohibited advertising material. But if you see any, please let us know.
27 February 2013
WDDTY #8 - Quantum Flapdoodle
Can quantum physics help sell pseudo science? More ASA adjudications against adverts in What Doctors Don't Tell You
 Harmony United Ltd.
Harmony United Ltd.
One of the hallmarks of pseudo science is its attempts to commandeer established words and phrases from science and misuse them for its own purposes, giving it a veneer of authority and respectability.
The prime example of this is the word 'quantum'. It refers to small, discrete steps such as the energy levels of an electron. In everyday use, however, it has come to mean the exact opposite: a quantum leap generally refers to some huge step in something or other.
But the word is certainly sciency-sounding and used by many purveyors of pseudo science, trying to give their product (there always is one) some legitimacy. The term 'quantum flapdoodle' was coined by Murray Gell-Mann in his book The Quark and the Jaguar: Adventures in the Simple and the Complex to describe such usage.
In the September and October issues of What Doctors Don't Tell You, there were full-page adverts for a website www.what.getinharmony.com. Here you will find all sorts of products including a Home and Office Harmoniser that, for £847.00:
Removes all health damaging influences.
In case that's not sufficient, you might need the Harmony Omega II with 'enhanced abilities' for a more respectable £2,897.00.
This website even has its own description of quantum physics — one no quantum physicist would endorse — imploring us that "quantum fields are God" and:
This is what Quantum Physics is - the rediscovery of who you are and the herald of a new beginning for mankind as he re-awakens her divine presence here on Earth.
The two adverts in What Doctors Don't Tell You advertised this website.
We questioned the implied claims about their products increasing health and performance and the Advertising Standards Authority (ASA) published their adjudication today.
In defence of their claims, the advertiser, Harmony United Ltd, said that their products:
…ensured that energy and information were sufficiently ordered so that the body and mind could utilise them. They said whether the human body utilised the higher quality order to remove disease was a matter for the human body. They pointed out that their technology had no influence on that.
No, we don't know either.
As the advertiser wasn't able to substantiate the claims, our complaint was upheld, with three breaches of the CAP Code for each ad.
 International Antiaging Systems Ltd
International Antiaging Systems Ltd
N-acetyl-5-methoxytryptamine (melatonin to you and I) is an unlicensed medicine in the UK, but is freely available in some other countries as a food supplement. It's a neurohormone produced in the pineal gland that helps regulate sleep-wake cycles.
An advert in September's What Doctors Don't Tell You was for a melatonin product, Melatonin Zn Se, that also contained zinc and selenium. According to the ad, these:
…work synergistically with melatonin to produce a much greater effect.
The ad proclaimed that the product could
Preserve Youthful Sleep Patterns
It is possible to be prescribed melatonin in the UK (and at least one form of of melatonin is licensed in the UK), but if it needs to be imported, this can only be done for an individual on prescription from a doctor, using an importer with an appropriate Wholesale Dealer's Licence. Members of the public may be able to import an unlicensed medicine for their own personal use, but it is clear that the advertising of unlicensed medicines in the UK is not permitted.
This ad claimed:
Melatonin Zn Se is freely available in Italy and is now available for the first time under article 1 of the treaty of Rome direct from Italy to you!
This is odd given that, according to the ASA, the advertiser is based in Belize in Central America, they have a PO Box on Sark in the Channel Islands, their domain name is registered in Panama and their parent company is also based in Panama, yet give a London contact phone number. The website mentioned in the ad, www.melatoninznse.com, is registered to an address on Sark and gives the same London telephone number.
Regardless, because it is an unlicensed medicine in the UK, it is prohibited to market it in the UK, regardless of where it comes from.
We challenged the ad and the ASA's adjudication on it is also published today.
Trying to defend the marketing of an unlicensed medicine, the advertiser said they believed:
…their ad was lawful, decent, honest and truthful, because they believed that individuals had the right to import medicinal products for their own use.
The ASA upheld our complaint, ruling it breached the CAP Code and told the advertiser not to market unlicensed medicines again.
As always, advertisers should note that adjudications don't just apply to the medium the ad was in, but to all marketing communications — claims are likely to be just as misleasing regardless of where they are published.
Adding up

With 52 CAP Code breaches in eight adjudications on ten adverts and complaints on eleven adverts informally resolved, we now have a further five still to be resolved.
There have now been several resolved complaints about supplements making claims that mentioned medical conditions in breach of the CAP Code and these advertisers now have an ASA adjudication against them or have been informally resolved.
We have already reported an advertiser of a medical device to the medicines regulator (the MHRA) after they refused to acknowledge the ASA as a competent authority. If other advertisers don't withdraw or amend adverts to make them CAP Code compliant, will further complaints have to be made?
13 February 2013
WDDTY #7 - Going to Ground?
Two further ASA adjudications on adverts in What Doctors Don't Tell You this week…
 Earthing
Earthing
Quoting that ever-reliable source of medical information, the Daily Mail, this advertiser in the first two issues of What Doctors Don't Tell You announced that many of us don't get enough sleep.
The solution? Go to ground. Earth yourself:
The Earth then shares it antioxidant, anti-inflammatory, anti aging electrons from its inexhaustible store. [sic]
The Earth also stores natural rhythms- day/night, and reconnection supports sleep.
The products are mats that you lie/sit/work on that are conductive and connect to the earth on a mains socket.
But if you think this might not be supported by the evidence, you needn't worry.
Do clinical studies support the claims made for Earthing?
Robust studies show significant improvements in sleep, vitality, rebalancing of key hormones (cortisol- the stress hormone, influences thyroid hormone), improvements in circulation and reductions in blood pressure.
Extensive case studies on reduction of inflammation (associated with any -itis medical condition such as arthr-itis) were accompanied by reduced sensation of pain.
Any -itis medical condition? Wow.
We thought the Advertising Standards Authority (ASA) might like to update their guidance to include these 'robust studies'.
We challenged the claims:
Sleep better tonight- sleep earthed
Earthing Institute Studies show:
100% of people woke feeling rested
85% of people fell asleep faster
93% experienced better sleep
78% experienced better well being
82% reported reduced muscle stiffness/pain
Sokal study reports benefits in blood sugar regulation, thyroid hormones, osteoporosis metabolic.
Sinatra study shows blood thickness reductions and circulation benefits.
Poor sleep can have a serious knock-on effect on your health
Robust studies show significant improvements in sleep, vitality, rebalancing of key hormones (cortisol- the stress hormone, influences thyroid hormone), improvements in circulation and reductions in blood pressure…Extensive case studies on reduction of inflammation (associated with -itis medical condition such as arthr-itis) were accompanied by reduced sensation of pain.
We also challenged whether the use of the title Dr in the name Dr John Kelsey PhD, ND was misleading as we understood that Kelsey did not hold a general medical qualification.
The ASA upheld our complaints and their adjudication is published today.
The advertiser, BEP Technology, trading as Original Earthing (BEP), claimed that they were not making medical claims but that they were promoting 'optimal health':
They said conventional medicine was concerned with [treating disease] and explained that natural resources and elements in the world were exempt from such considerations because they dealt only with optimising health.
However, they still provided the ASA with studies, articles and case studies in support of their claims.
The advertiser claimed that:
…the readership of WDDTY were a specific group with a demonstrated interest in health and who were naturally sceptical with a good, discriminatory awareness and who were therefore unlikely to be misled by any claims in the ad.
Ground breaking
The ASA considered that many of the claims being made for the efficacy of earthing would be 'ground breaking'! As such, they would require a high level of substantiation:
…such as a body of scientific research including clinical trials conducted on people
Since the advertiser had not supplied this, the ASA ruled that the claims were misleading and that some claims were for medical conditions for which medical supervision should be sought and that:
…advice, diagnosis and treatment for them should only be conducted under the supervision of a suitably qualified health professional. Because we understood that was not the case in this instance, we concluded that those claims breached the Code as they could discourage consumers from seeking essential treatment for those conditions.
The ASA were not at all impressed with the advertiser's argument that Kelsey's PhD in Process Engineering entitled him to use the title Dr in the context of an ad making medical claims and ruled it misleading.
This ad breached the CAP Code on five counts.
For further information on earthing and how it is being marketed, read Eric Hall's article on Skeptoid: More Mercola Misinformation: Grounding In Reverse.
 When Antibiotics don't Work
When Antibiotics don't Work
Yet another supplement.
This is the first of three adverts in What Doctors Don't Tell You by Dulwich Health Ltd we complained to the ASA about. This one was for a product called AlliTech that stated that it was:
…nature's powerful anti-biotic, anti-parasites, anti-viral, anti-fungal and immune booster with no side-effects reported during over 10 years of use.
The ad made many claims for its effectiveness for several conditions including pneumonia, upper respiratory infections, Lyme disease, kennel cough and, of course, dysentery in apes. It also claimed that 52 patients at University Hospital [sic], London fully recovered from long term MRSA after taking allicin, one of the ingredients of AlliTech. And it has saved thousands of Horse Chestnut trees from dying from Pseudomonas Syringoefungus [sic].
Impressive claims for a product derived from garlic.
We challenged the claims made; whether doctors did indeed believe that parasites were the most under-diagnosed and under-appreciated health threat that was linked to Alzhiemer's, dementia, Parkinson's and MS; whether the testimonials were genuine and whether the ad made claims for an unlicensed product.
The ASA's adjudication is also published today, upheld on all four issues.
The advertiser acknowledged receipt of the ASA's letter and said they noted their comments.
They made no attempt to justify or substantiate the claims they made, so the ASA ruled that they had breached the CAP Code on five counts and told them not to repeat the ad, not to make efficacy claims unless they could be substantiated and not to make medicinal claims for an unlicensed product.
The Tally
With 45 CAP Code breaches in six adjudications on seven adverts and complaints on eleven adverts informally resolved, we now have a further eight still to be resolved.
And the February issue What Doctors Don't tell You has sixteen fewer pages than the previous five issues.
06 February 2013
WDDTY #6 - The Missing Link?
Another informally resolved case to add to the ever-growing list
 Q-Link Clear
Q-Link Clear
Combat stress, poor performance and fuzzy thinking; fight electronic stress. Proven science; published peer reviewed papers. Peace of mind; immediate results.
Electromagnetic fields from electronic devices can, apparently, undermine performance and well-being and have a biological effect on the body.
So any device that protects us from these must be a good idea, whether it uses 'Sympathetic Resonance Technology (SRT™)' or not. The missing link in our hectic lives?
This is yet another ad placed in both the September and October issues of What Doctors Don't Tell You that we submitted to the Advertising Standards Authority. The advertiser is Clarus, based in California (but whose products are available all over the world) advertising their Q-link Clear,
Their website claims:
The Q-Link CLEAR is a new and exciting addition to the well-known Q-Link range of "body" products. Programmed with over 100 natural frequencies known to support the biofield, the Q-Link CLEAR is compact and feather-light so it won't get in the way. Just attach it to a device where it will come closest to your body. Then, when you use your mobile phone, or listen to music on an mp3 player, hold your game player or otherwise - the Q-Link CLEAR focuses and strengthens the biofield's ability to support your body's natural defenses to EMFs and other stressors.
According to their website, SRT™ is:
Sympathetic Resonance Technology™ (SRT™) is based on the fundamental scientific discovery that every physical system has fields of energy that permeate and surround that system. When systems vibrate at their optimal frequency spectrums, they are able to function more efficiently and deliver expected, even enhanced, levels of performance. When these systems are not vibrating ideally, reduced efficiency and performance can result. In humans, the principal energy system is sometimes referred to as the “Biofield” – a term recognized by the U.S. National Institutes of Health.
SRT™ is an array of proprietarily identified frequencies that support and enhance the efficiency and performance of various organic and inorganic systems. Biological, electrical, chemical and other physical systems influenced by SRT™ applications exhibit increased functionality, coherence, structural integrity and other positive characteristics and benefits
And their trade mark for SRT™ appears genuine, covering:
Treatment of materials by application of electromagnetic energy in the nature of subtle energy frequencies in order to reduce quantum noise and increase the efficiency of energy exchange for the purpose of improving the functioning or extending the useful life of the materials.
So now we know.
But for an alternative view on SRT™ technology and what's in a q-link pendant — which uses the same SRT™ technology — read Ben Goldacre's The Amazing Qlink Science Pedant.
Their ad claimed that it was 'proven science' and there was 'published peer reviewed papers' and their website claims SRT™ has 'proven its effectiveness in a wide range of experiments, including trials at UC Irvine, University of Vienna, and Imperial College London'. However, for some reason, they agreed with the ASA not to repeat efficacy claims until they have sufficient evidence to comply with the CAP Code and assured the ASA that the ad would be withdrawn and amended.
This empathises that holding evidence just isn't enough; it has to be evidence that meets the requirements laid down in the ASA's guidance on substantiation.
Because it was informally resolved, Clarus gets their name published on the ASA's website today as an informally resolved case.
If they had chosen to try to substantiate their claims, the ASA might have formally investigated and examined their evidence — I wonder what that would have found?
Counting
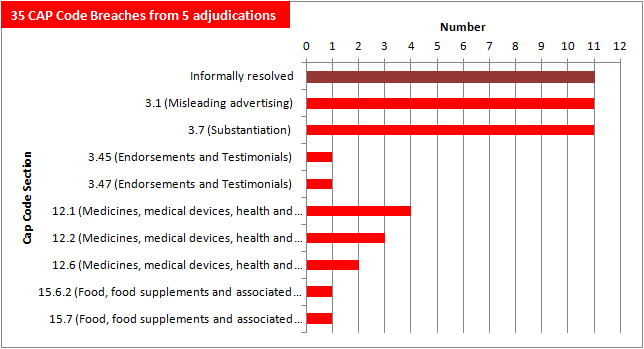 Still an impressive 35 CAP Code breaches from just five adjudications plus eleven informally resolved cases — with many more to come.
Still an impressive 35 CAP Code breaches from just five adjudications plus eleven informally resolved cases — with many more to come.
Programmer
Thanks to all who offered their services and we'll be in touch with everyone shortly.
23 January 2013
Latest news
- "Undisputable evidence of scientific misconduct" by homeopaths
- Yet another bad year for homeopathy
- Nelsons Homeopathic Pharmacy #3
- Nelsons Homeopathic Pharmacy #2
- The Society of Homeopaths: failing to make the case for homeopathy
- The end of homeopathy on the NHS in Bristol?
- NHS Homeopathy: 20 years of decline
- The different faces of the Society of Homeopaths
- The growing pains of osteopaths
- Diluting misleading claims - ASA update
Most read
- Finding deleted and changed webpages
- About The Nightingale Collaboration
- How to find out who owns a website
- Advertising Standards Authority
- Rubbing salts into the wounds of homeopathy
- How to submit a complaint to the ASA
- The decline of homeopathy on the NHS
- Landmark decisions for homeopaths
- NHS Lanarkshire to end referrals to Glasgow Homeopathic Hospital
- Making a complaint



