News
News items will be published here, but also sign up to our Newsletters — see the form on the right.
You can receive updates from the News feed, either to your RSS reader or via email.
Rubbing salts into the wounds of homeopathy
Homeopathy has suffered many body blows recently. A new decision by the medicines regulator rubs salt into its wounds.
The last year or so has not been good for homeopathy. The comprehensive Australian National Health and Medical Research Council concluded that "there are no health conditions for which there is reliable evidence that homeopathy is effective." The FDA and the FTC in the US are considering their positions on the regulation and advertising of homeopathic products and will hopefully clamp down on the worst excesses of misleading claims made there. In the UK, homeopathy continues its downward spiral, it's being challenged in Liverpool and the Department of Health will consult next year of the complete blacklisting of homeopathy. 2016 is going to be a very interesting year for homeopathy.
Last August, we published details of our complaints against Nelsons Homeopathic Pharmacy. We're still waiting for the General Pharmaceutical Council to complete their investigation, but the medicines regulator, the MHRA, has already published their decision. In that newsletter, we said we were waiting for a further response from the MHRA. Although we're still waiting for a formal reply from them, the essence of what we were waiting for has just been published by them in yet another complaint about homeopathic products.
Schuessler Salts
Wilhelm Heinrich Schüßler (Schuessler) was a German medical doctor and naturopath. He concocted twelve biochemic 'cell salts' or 'tissue salts' (as he called them), intended to redress percieved bodily deficiencies in one mineral or another. Frequently referred to as 'microdoses' these are accepted as homeopathic by some homeopaths and rejected by others. When I asked one manufacturer, I was told:
Tissue Salts are not homeopathic but are homeopathically prepared micro dose minerals. Dr. Schuessler believed that it was most effective to follow the principles of homeopathy in their preparation – 1 part mineral to 9 parts lactose – mortar and pestle for 20 minutes bringing it up to a 1X and so on. They can be touched by hand and work differently in the body navigating all that you eat and ingest where they need to be. They are not as sensitive as homeopathics. They can be taken alongside food, coffee etc.
And:
Tissue Salts are not Homeopathics but are micro dose minerals that follow the similar method used in preparation - they will not interfere with homeopathic Nat Mur - it is important to note that Minerals are not homeopathic, they are biochemic. They do not follow the rule of similarity as many consider but are prepared in the homeopathic manner in order to provide bioavailability. The system needs minerals in order to function in these minute doses. They are absorbed through the mucosa that enables them direct entry to the blood stream and the availability to the cells.
At least that's clear…
Whatever the different factions within homeopathy believe, Schuessler Salts are frequently labelled and sold as being homeopathic. But are they as ineffective as homeopathy? The Commissioner for Public Health in Australia looked into one of these salts and declared:
THE following report is issued under section 210 of the Health Act, 1911-1944:—It is claimed that the above "remedy" [Dr. Schuessler's Cell Salts, Kali Phos. 3X] is "indicated in loss of mental power, brain fag, paralysis of any part, nervous headaches, neuralgic pains, general disability and exhaustion and sleeplessness from nervous disorders." The "remedy" has been analysed and been found to contain potassium dihydrogen phosphate and lactose. The actual quantity of potassium dihydrogen phosphate in the "adult dose" is so minute that over 9,000 tablets would be necessary to give the minimum medicinal dose of this drug. Lactose is a sugar which is of no value in the treatment of any of the above-mentioned maladies. Dr. Schuessler's Cell Salts can therefore have no curative value. They will bring about no improvement in any of the illnesses for which they are said to be indicated. Any expenditure on the purchase of these salts will be money wasted.
— C. E. COOK, Commissioner of Public Health
That was in 1946.
One of the main brands of these products is an Australian company, Martin & Pleasance, but there are others, including the New Era range (once owned by the pharmaceutical giant, Merck through their Seven Seas brand, but now owned by the Italian company, Olimed Ltd, sold in the UK by Power Health Products Limited).
Nelsons
It was the Martin & Pleasance products we found being advertised on Nelson's website. Because they do not have a licence under the National Rules Scheme, an authorisation under the Homeopathic Rules Scheme nor even a (defunct) Product Licence of Right, we questioned whether these could be unlicensed medicines. The significance is that if the MHRA ruled they were unlicensed medicines, it would be a breach of the Human Medicines Regulations 2012 (HMR 2012) to advertise, supply or sell them.
 Today, the MHRA published their decision on a complaint about the advertising of these or similar products by Homeopathy Express, Nutricentre and Health Stuff. Although these were not our complaints, the MHRA have now published what they told us previously about Schuessler products and what we've been trying to get them to state publicly:
Today, the MHRA published their decision on a complaint about the advertising of these or similar products by Homeopathy Express, Nutricentre and Health Stuff. Although these were not our complaints, the MHRA have now published what they told us previously about Schuessler products and what we've been trying to get them to state publicly:
MHRA guidance: Tissue Salts/Schuessler Salts are considered to be medicinal products if any of the following appear in promotional material:All other cases will be reviewed on a case-by-case basis.
- the terms ‘homeopathic remedy’, and/or ‘homeopathy’ and/or ‘homeopathically prepared’
- a numerical value for the potency eg 6X and/or the word ‘potency’ itself
- indications for use in a medical condition
So, it's perfectly OK to advertise these products as long as it is not claimed that they are in any way homeopathic, they do not have a homeopathic 'potency' and they have no therapeutic indications. If they comply with all of these restrictions, they will not be medicines, but will simply be food supplements and have to comply with food regulations. This is essentially the same as when Bach Flower products were declared to be foods, not medicines nearly a year ago.
The standard labelling of these products (such as the one in the photograph) stated they were '6X' 'potency' and the mere mention of this is sufficient to bring it within the MHRA's definition of a homeopathic medicine. This is a direct consequence of the EU Directives and the HMR 2012: homeopathic 'medicines' are regulated and anything that is presented as being homeopathic is a medicine and has to comply with these rules.
If they are not presented in any way as homeopathic, then they are just foods, not medicines.
 Quite rightly, the MHRA will review each product individually, but we think it's clear that this ruling applies to the following:
Quite rightly, the MHRA will review each product individually, but we think it's clear that this ruling applies to the following:
There may be others and if you spot any on sale in the UK that appear to be contravening the MHRA's new guidance, please let us know and consider submitting a complaint to the MHRA about them.
The aftermath
Before our complaint against Nelsons, their website said:

That page now says:

There is now no mention that these products are homeopathic.
But what about the websites of the three sellers mentioned in the MHRA's decision?
Homeopathy Express
This is advertising New Era Calc Sulph (No.3) Tissue Salts and Brittle Nails & Falling Hair (Comb - K), but both are currently not available and another page says they have been discontinued. It looks like they also used to sell the Dr Reckeweg brand of tissue salts but they have been removed.
Health Stuff
Whatever Schuessler/cell/tissue salt products they sold, they are no longer advertised on their website.
Nutricentre
Thirteen of the sixteen Schuessler products they advertise are currently out of stock.
For two of the three that are available, the description shows:
Congestion by Schuessler Tissue Salts. Kali Mur Nat Mur & Ferr Phos For relief from symptoms associated with Chest congestion Coughs Colds Available in tablets. Always read the label use only as directed. If symptoms persist consult your healthcare professional. For over 150...
Schuessler Combination H Tissue Salt is a complete natural formula for hayfever and allied conditions with mag phos nat mur and silica. Hayfever is an allergic condition of the mucous membranes of the nose eyes and upper respiratory tract and is common during spring when sensitivity to pollen is severe but it is very treatable by combination H particularly if treatment is started from six weeks before the expected onset of normal symptoms.
For Calc Fluor No 1, although out of stock, the product page states:
The cell salt Calcium fluoride is primarily a salt of the connective tissue
Also the bones and parts of the skin and blood vessels belong to the connective which is why Calcium fluoride can be used for all problems of these tissues
…
Ingredients: Calcium Fluoride 6X Lactose Monohydrate Calcium Hydrogen Phosphate Magnesium Stearate
Many of the others have similar information.
These would appear to be therapeutic indications and some refer to the potency as 6X, possibly contravening the MHRA's guidance so we'll be bringing these to their attention and hope that they will be corrected before the products return to stock.
This is another step forward for informed consumer choice, but another blow for homeopathy. Gradually, sellers of homeopathy and homeopathy products are being held to account. Homeopathy is taking a beating, perhaps fatally wounded.
Addendum
I have just come across this advert for New Era Tissue-Cell Salts in the February 1951 issue of the homeopathy magazine, Heal Thyself:
It's a pity the Advertising Standards Authority wasn't around then.
30 December 2015
On a downward spiral

Not that long ago there were five homeopathic hospitals in the UK: London, Tunbridge Wells, Bristol, Liverpool and Glasgow. The Tunbridge Wells and Liverpool hospitals have since closed and the pharmacy at the Glasgow one is no more.
As for the Bristol hospital, it started out in its own building near the centre of Bristol (Cotham Hill), then it became a small clinic in the new South Bristol Community Hospital. Earlier this month, just three years later, this too closed to be replaced by a private clinic, the Portland Centre for Integrative Medicine, that is no longer part of the NHS but that is now just contracted to provide homeopathy services to University Hospitals Bristol. It's situated in Litfield House Medical Centre, which offers private consulting rooms, some six miles from the hospital.
And then there were two
Today, there remains only the Royal London Hospital for Integrated Medicine (the re-named Royal London Homeopathic Hospital) and the Glasgow Homeopathic Hospital (aka the Centre for Integrated Care, as they like to call it).
NHS Lanarkshire was the latest of the Scottish health boards to end funding of all treatments at the Glasgow Homeopathic Hospital, and, in August, one of it's previous patients from Lothian lost her judicial review at the Court of Session of the earlier decision by NHS Lothian, finally putting an end to the homeopathy supporters' protracted battle.
But now we know that even the jewel in the homeopaths' crown, the RLHIM, no longer has a homeopathy service.
We've known this for some time as it was admitted by them in their response to a Freedom of Information Act request last year and this service has not been mentioned on their website for some time. It is now public thanks to someone else's FOIA request. All they provide now are:
- Acupuncture Services
- Adult Allergy Service
- Allergy Service
- Chronic Fatigue Service
- Children’s Service
- Complementary Cancer Care Service
- Fibromyalgia Syndrome Service
- General Medicine Service
- Hypnosis Service
- Insomnia Service
- Irritable Bowel Syndrome Service
- Musculoskeletal Medicine Service
- Nutrition and Dietetics
- Podiatry Service
- Rheumatology Service
- Skin Service
- Psychological Services
- Weight Loss Service
- Women’s Service
But no homeopathy service. For their flagship hospital, closing that down must have been a bitter sugar pill to swallow.
Even though homeopathy isn't mentioned they still provide it under the guise of other services. The list of services doesn't match those listed on their website (and there are some interesting differences), but some do include homeopathy:
Of course, we've won several Advertising Standards Authority rulings and informally resolved cases against the RLHIM over claims they made on their website and in leaflets. In Homeopathy clinic toes the line, we briefly mentioned Prof David Colquhoun's blog post: Conflicts of interest at the Homeopathic Hospital. It's good to see that the RLHIM has since made this clearer:

And in overall terms, we also already know that the number of prescriptions for homeopathy products supplied by community pharmacies in England has fallen by 94% in the past 17 years.
We also know that very few Clinical Commissioning Groups fund homeopathy.
How much longer will NHS homeopathy survive this downward spiral?
Ainsworths
After Nelsons were reprimanded in August (as a result of our complaint), another homeopathy manufacturer was today admonished by the medicines regulator, the MHRA.
As part of MHRA’s regular review of advertising, we reviewed the Ainsworths website. Some unlicensed homeopathic remedies had the name of a commonly recognised disease or medicine. We were concerned that this could be seen as a claim to treat or prevent that disease. We were also concerned that remedy kits containing unlicensed remedies were being promoted for sale in the UK.
Ainsworths agreed to remove the name of the disease and the medicine names from unlicensed remedies on their price list and amend the web pages with remedy kits.
It's encouraging to see that the MHRA instigated this themselves rather then waiting for a complaint from a member of the public or a pharmacist, but it's disappointing that they have not given details of what products they were concerned about.
In terms of the kits, they have been in trouble before, so it's disappointing to see them being pulled up again, apparently for exactly the same breach of the medicines regulations.
Note that the decision is dated 10 June, yet the decision was only published today. It's therefore even more disappointing and concerning to see Ainsworths still advertising unlicensed remedies such as these:
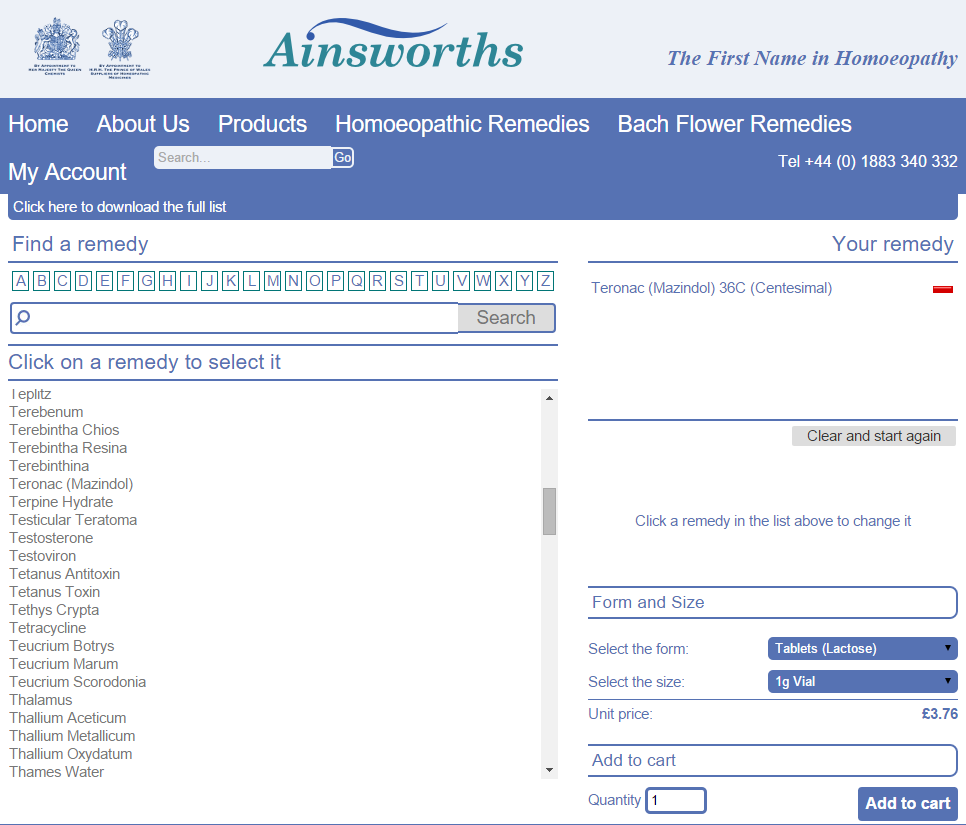
We sincerely hope that Ainsworths will comply with the MHRA's instructions to remove all products with the name of a commonly recognised disease or medicine, and — in the interests of not misleading the public — products that have names very similar to commonly recognised diseases or medicines. We're sure Ainsworths would not want to mislead consumers but we hope the MHRA will continue to monitor their website and take further action as necessary to protect consumers.
Photo credit
The defunct Tunbridge Wells Homeopathic Hospital by Tannice Hemming.
22 October 2015
Nelsons Homeopathic Pharmacy #1
The medicines regulator, the Medicines and Healthcare products Regulatory Agency (MHRA) have today published their decision on a complaint we made to them a few months ago about the homeopathy manufacturer and seller Nelsons Homeopathic Pharmacy.
As usual, the MHRA give few details of the complaint, their investigation, what they found or what they decided:
Even though there is a link that purports to give more information, this simply links to the page where this decision is listed along with others for July. They even fail to give the proper name for the trader, Nelsons Homeopathic Pharmacy, or give the urls of the two websites involved: http://www.nelsonsnaturalworld.com and http://www.nelsonspharmacy.com/.
It's not clear to us why these decision notices are so void of any details that would help consumers: the Advertising Standards Authority (ASA) by contrast, when they publish an adjudication, give full details of the complaint, their investigation, the advertiser's response, their decision and the sanctions applied. For example see this ASA adjudication against another homeopathy manufacturer, Ainsworths.
The issues we highlighted to the MHRA were mostly about the advertising of homeopathic products that was not within the terms of the authorisation or registration for those products. For example, their Arnicare Arnica 6C product stated:
Indications: For the symptomatic relief of sprains, muscle aches, bruising and swelling after contusions.
However, this is a Homeopathic Rules (HR) scheme product and its registration does not permit therapeutic indications. Instead, all advertising for HR products must simply contain the text:
A homeopathic medicinal product without approved therapeutic indications.
It's a moot point whether the general public understands this to mean that there is not a jot of good evidence that these products have any therapeutic effects whatsoever, a point raised by the House of Commons Science and Technology Select Committee in their Evidence Check on homeopathy.
Another page advertising their Aconite 30C National Rules (NR) scheme product contained the following instructions:
They can even be dissolved in warm water if preferred.
However, the Public Assessment Report for ths product gives the posology and method of administration as:
Adults and children: Take 2 pillules every 2 hours for the first 6 doses, then 4 times daily until symptoms improve for up to a maximum of 7 days.
Pillules should either be chewed or placed under the tongue until dissolved.
Although it makes not the slightest difference to the 'effectiveness' of the homeopathic product, there is no mention of dissolving in warm water as a permitted method of administration.
The other issues covered by this decision were similar to these and we suspect they were simply oversights by Nelsons — they have now corrected them.
In total we identified eight issues with the advertising of their products on their websites, including the ones above. When the MHRA told us of their decision, we queried a couple of points and we're waiting for a further response from them. We'll let you know when these have been satisfactorily resolved.
However, the issues the MHRA have dealt with were just part of our larger complaint to both the MHRA and the General Pharmaceutical Council (GPhC), the statutory regulator for pharmacies and pharmacists. Once the GPhC have completed their investigations into all the other issues we raised, we'll let you know.
Meantime, we've added this MHRA decision to our growing list of published results.
14 August 2015
Stemming the tide
The list of misleading adverts in the magazine What Doctors Don't Tell You sometimes seems endless,
 The April 2015 issue of What Doctors Don’t Tell You carried an advert by Life Long Products for their 'Stem Cell Therapy (SE2)' product that claimed it could improve the body's ability to:
The April 2015 issue of What Doctors Don’t Tell You carried an advert by Life Long Products for their 'Stem Cell Therapy (SE2)' product that claimed it could improve the body's ability to:
- MAINTAIN optimal health
- BOOST your immune system
- PROTECT your nervous system
We know that boosting the immune system is a common claim made by advertisers of alternative therapies and supplements and something they might like us all to think is good for us. In fact, it turns out that boosting the immune system is not such a good idea at all.
The testimonial in the advert could also give the impression that arthritis sufferers using this product can stop using painkillers.
As has been noted elsewhere, there is frequently a connection between adverts and articles, and this one boasts:
StemTech as featured in WDDTY Jan & March Issues - by Vet Paul Boland
Can stem cell therapy provide these benefits? The product page on the advertiser's website states:
Stemtech’s SE2® is the world’s first all-natural supplement documented to support the release of adult stem cells from bone marrow. Our advanced supplement puts more stem cells in the bloodstream, and the effect lasts longer.
An advancement in Cellular Renewal – helping Nature do what it is designed to do
This 'advanced supplement' doesn't come cheap, of course: a bottle of 60 capsules costs £60.95 (plus shipping), but it might seem a bargain if the claims stood up to scrutiny.
But this product isn't regulated by the Human Fertilisation and Embryology Authority or even the Medicine and Healthcare products Regulatory Agency (MHRA): it isn't stem cell therapy derived from bone marrow, umbilical cord blood, etc, but simply a food supplement.
As a food supplement, the only claims permitted in advertising are those listed on the EU Register of nutrition and health claims and in the UK, those advertising claims are regulated by the Advertising Standards Authority (ASA).
Because we didn't believe the claims being made were authorised claims, we submitted a complaint to the ASA.
While we were at it, we looked further at the advertiser's website and included complaints about some of their other products: Jamu (cached), Forever Freedom® Aloe Vera Juice (cached), Serrapeptase (SerraEnzyme)- High Potency Capsules (cached) and PainSolv MkV Class IIa Medical Device (cached).
The page for the Jamu product no longer exists and the advertiser appears to have completely removed it from their website, but they had claimed:
Jamu (formerly Djamu) is traditional medicine in Indonesia. It is predominantly herbal medicine made from natural materials, such as parts of plants such as roots, bark, flowers, seeds, leaves and fruits. Materials acquired from animals, such as honey, milk, Ayam Kampung eggs and goat’s bile, are also often used.
…and they listed more than a dozen products in the range with descriptions such as 'Cholesterol Control', 'Naturally Lowers Blood Pressure', 'Naturally Dissolves Kidney Stones' and 'Naturally Reduces Blood Glucose Levels'.
ASA investigation
The ASA originally said they would contact Life Long Products and ask for their comments on several points they identified in the website advert for PainSolv references to pain reduction and management; some of the claims were made in a video on their website, but which is also within the ASA's remit. The advertiser could then provide the required evidence to substantiate the claims or agree with the ASA to remove the claims.
For the other points of the complaint about the supplements and Jamu herbal product, the ASA said those were sufficiently straightforward and would take them up informally with the advertiser, asking them to:
…remove the health and disease claims from their Stem Cell Therapy, Aloe Vera and Serrapeptase, and to remove the Jamu products from their marketing activity.
Although not stated, we believe the Jamu products were unlicensed medicines that would be a breach of the Human Medicines Regulations 2012 to advertise, supply or sell to the public and we're pleased to see that there is now no reference to these products on the advertiser's website.
Serrapeptase has, of course, been the subject of a previous complaint of ours against an advertiser in What Doctors Don't Tell You. That time, the ASA adjudicated on the evidence Good Health Naturally provided but ruled it was not adequate to substantiate the claims made. They upheld our complaint on all four points, identifying eight breaches of their CAP Code.
However, instead of providing evidence to substantiate their claims for PainSolv, the ASA notified us that the advertiser had agreed to:
…implement suitable changes to their website to bring it in line with the CAP Code.
So, instead of an adjudication, the ASA have today published the outcome of our complaint as an informally resolved case.
Except...
Except… although the advertiser has removed all mention of their Jamu products from their website, we see no other changes as yet.
If those pages don't change soon, we'll bring them to the attention of the ASA.
HealthWatch UK
 The charity HealthWatch (not to be confused with the recent NHS organisation "Healthwatch England") has a study under way to test the effectiveness of consumer protection laws against misleading health claims.
The charity HealthWatch (not to be confused with the recent NHS organisation "Healthwatch England") has a study under way to test the effectiveness of consumer protection laws against misleading health claims.
A previous study by HealthWatch, Spurious Claims for Health-care Products: An Experimental Approach to Evaluating Current UK Legislation and its Implementation found some worrying results showing widespread variation in the application of consumer protection law and a reluctance to enforce it to protect consumers. Now is the time to build on that research with this bigger study.
Useful data are coming in, but more volunteers are urgently needed.
The study comprises these steps:
- Request traders for evidence to support claims made on their websites. You will be provided with trader details, 5 for each volunteer.
- Ask traders to stop making false claims.
- Complain to Trading Standards, via the Consumer Direct website.
- Follow up each complaint for six months.
All data are captured in a suite of online forms, and results will be submitted to a major journal.
If you are interested, please contact HealthWatch trustee Les Rose. Although it's not at all labour-intensive (so the current team reports), please only volunteer if you have the time.
12 August 2015
Another WDDTY advertiser in hot water
Our last sojourn into adverts in the monthly magazine, What Doctors Don't Tell You (WDDTY) concerned claims about Treating Ebola with 'bioresonance'.
 This time, we submitted a complaint to the Advertising Standards Authority (ASA) about an ad by Water for Health Ltd in the December 2014 and January 2015 issues and the advertiser's website. In their adjudication published today, the ASA identified two points and upheld them both.
This time, we submitted a complaint to the Advertising Standards Authority (ASA) about an ad by Water for Health Ltd in the December 2014 and January 2015 issues and the advertiser's website. In their adjudication published today, the ASA identified two points and upheld them both.
Because of the number and extent of the claims being made, the points relate to those claims made on the advertiser's website, but the ruling applies to all media where the same claims are made — including WDDTY ads, of course.
Not just any Water…
The WDDTY ad and their website made several claims about the health properties of water: not just any water, but 'healthy alkaline ionised water'. The website claimed:
Alkaline nutrition has tremendous therapeutic power and is also vitally important to support your body if you are undergoing conventional medical treatment.
Their website also contained claims for a number of food supplements including chlorella, chia seedsia seeds, organic flax seeds oil, Brocco forte (a broccoli extract), organic prunes and 'maximised turmeric curcuminoids.
The claims we complained about included:
Green Food Supplements contain the nutrients to help to:
- Promote better body alkaline balance
- Energize the body from cellular level
- Remove toxins
- Improve immunity
Many of our natural health foods are also thought to play a significant part in:
- Improving the permeability of your cell membranes
- Promoting the health of your eyes, brain and skin
- Boosting your immune system
- Increasing resistance to a number of degenerative conditions
Chlorella has five main benefits:
- Promotes healthy cellular growth and repair to slow down aging
- Supports and strengthens the immune system
- Is highly alkaline to support a healthy pH in the tissues, and cells.
- Detoxifies and helps purify the body
- Provides complete nutrition to normalize body function
Chia Seeds are thought to help you:
- Support heart health
- Stabilize blood sugar
- Improve brain power
- Enable detoxification
- Encourage weight loss
[Organic Flax Seed Oil] will help to:
- Keep your skin, nails and hair healthy
- Maintain your cardiovascular health
- Ensure that your eyes and brain develop properly
- Boost your immune system
- Improve the flexibility, and permeability, of cell membranes, allowing for better cell hydration
Furthermore, taking an omega 3 Supplement like one of Omega Nutrition’s flax oils is recommended by several natural cancer treatment protocols, including the Budwig program and Gerson Therapy, and recent research also suggests that they may play a part in preventing neurological diseases such as Bipolar disorder.
Broccolo [sic] forte can help you to to [sic]:
- promote improved liver function
- combat allergens
- remove toxins
- neutralize toxic, cancer-promoting hormone metabolites
- only does it [sic] have a high broccoli sprout extract content, it is combined with 6 other immune boosting plant extracts to further boost its effectiveness.
The phytonutrients in organic prunes are also thought to help neutralize many of the harmful oxygen free radicals implicated in the development of:
- Cardiovascular disease
- Breast cancer
- Prostate cancer
- Alzheimer's disease
- Gastric Cancer
- Chronic fatigue syndrome
And some testimonials:
“I love the water. My skin is healing in front of my eyes.”
“All of the above have enabled me to reduce my prednisolone (steroids) with no difficulties and I can now see the end of their use within the next few months. My health has been turned around.
The ASA listed many of these and condensed them down to two points of complaint:
The Nightingale Collaboration challenged whether:
- the claims for the food and food supplements complied with the Code; and
- the claims related to alkaline water also complied with the Code.
Water for Health told the ASA that although they disagreed with the issues raised they had revised their advertising.
In their assessment, the ASA cited EC Regulation 1924/2006 on Nutrition and Health Claims made on Foods and pointed out that they were part of their CAP Code, saying:
…only health claims that appeared on the list of authorised health claims (the EU Register) could be made in ads promoting foods and that marketers must also ensure that they met the conditions of use associated with the claims in question. Health claims were defined as those that stated, suggested or implied a relationship between a food, or ingredient, and health.
The Regulation stated that references to general benefits of a nutrient or food for overall good health or health-related wellbeing were acceptable only if accompanied by a specific authorised health claim. Although some claims had been revised, we considered that they retained their original meaning. For example but not limited to, the revisions to the claims for Maximized Turmeric Curcuminoids, Clean Chlorella and Brocco Forte, which we considered were likely to be understood as general health claims, were not sufficient to alter their original impression.
We also considered that in the context of the ad, which included no reference to specific types of detoxification, "enable detoxification" for Chia Seeds, "Detoxifies and helps purify the body" for Clean Chlorella and "remove toxins" for Brocco Forte were likely to be understood to be general health claims. We noted that they were not accompanied by a related specific authorised health claim and were therefore not acceptable.
The ASA detailed the issues they had with the claims made and concluded:
Because the ad made general health claims which were not accompanied by a related specific authorised health claim, and included specific health claims, and a reduction of disease risk claim, for which evidence had not been provided that they were authorised on the EU Register, and because it included prohibited claims that the advertised foods could prevent, treat or cure human disease, we concluded that it breached the Code.
Pretty damning. The ad therefore breached CAP Code rules 15.1, 15.1.1, 15.2 and 15.6.2 (Food, food supplements and associated health or nutritional claims).
The second point of the complaint was about the claims for alkaline water. The ASA ruled:
Although Water for Health had told us that their ad had been revised, claims for "Alkaline Nutrition" continued to make the same claim that it "has tremendous therapeutic power and is also vitally important to support your body if you are undergoing conventional medical treatment". We considered in the context of the claims for the product's purported benefits for the body, the reference to alkaline nutrition's "tremendous therapeutic power" and that it was "vitally important to support your body" were likely to be understood as a general health claims, which we noted were not accompanied with a related specific authorised health claim as required by the Regulation. Because these general health claims were not accompanied by a related specific authorised claim, we concluded the ad breached the Code.
The ad therefore breached CAP Code rule 15.2 (Food, food supplements and associated health or nutritional claims).
Overall, the ASA ruled:
The ad must not appear again in its current form. We told Water for Health to ensure they did not make general health claims which were not accompanied by a relevant authorised health claim, or to include specific health claims or reduction of disease risk claims which were not authorised on the EU Register, or make claims to prevent, treat or cure human disease for foods.
So another misleading WDDTY advertiser.
Although the adjudication doesn't mention the ad in WDDTY explicitly, as always, the adjudication covers all adverts regardless of the medium: after all, if a claim is misleading in one medium, it will be just as misleading in others.
The latest tally
With this one, we now have 11 adjudications and 12 informally resolved cases against WDDTY advertisers and at least one of these advertisers — and WDDTY regular — Wholistic Research, has now ceased trading:

Guidance
The ASA provide several pages of guidance on the rules for health and nutrition claims for foods including:
These rules stem from the European Commission Regulation No 1924/2006 — the various authorised and non-authorised claims can be checked in the EU Register on nutrition and health claims.
(The ASA also helpfully provide a (non-exhaustive) list of some 444 Acts and regulations affecting advertising.)
The Government also produce Guidance on nutrition and health claims on foods, which states:
The ASA is the UK body responsible for advertisements in broadcast (TV and radio) and non-broadcast media. There are two advertising content codes: the Committee on Advertising Practice writes and maintains the non-broadcast advertising code (the CAP code) and the broadcast committee of advertising practice writes and maintains the TV and radio advertising standards code (the BCAP code). The ASA is the independent body responsible for administering those codes and is able to require advertisers and broadcasts to remove non-compliant claims. The advertising codes reflect the requirements of the Regulation.
With all this advice and guidance, there should be no need for any advertiser to make non-authorised nutrition and health claims for foods. Additionally, the ASA offer free advice to advertisers via their Copy Advice Team as well as other help to prospective advertisers.
No excuse
If Water for Health weren't aware of authorised and non-authorised claims under the Regulations, they are now. We welcome their decision to amend their advertising even if that initially did not go far enough to satisfy the ASA. They have made other changes, but we don't think it yet complies and we hope that they will look again at the claims they made for the products mentioned in the ASA's adjudication and ensure that they are now compliant.
They might also like to check the claims they make about the other products they advertise — whether on their website or in the pages of What Doctors Don't Tell You.
There can be no excuse for not complying with the rules, regulations and laws in place to protect the public.
27 May 2015
The (further) decline of homeopathy on the NHS
As yet another World Homeopathy Awareness Week starts, we should be aware that it appears to be in terminal decline in the English NHS
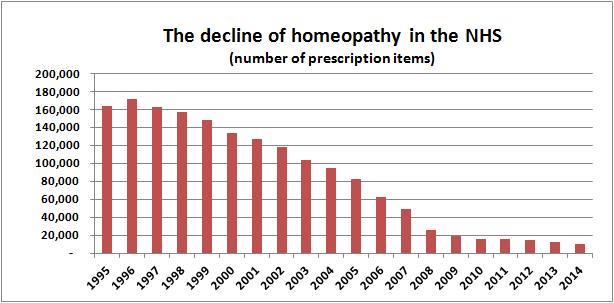
A year ago, we reported on the decline in the number of English NHS prescriptions for homeopathy that were fulfilled in community pharmacies. This showed a steady decline over the previous 17 years, down to just 7.5% of the number in 1996, with each year fewer than the previous.
These data come from the Prescription Cost Analysis data for England provided by the Health and Social Care Information Centre (HSCIC), the official source of data on the NHS.
My blog post, An idiot’s guide to understanding NHS homeopathy prescription data, gave the details of where these data came from and how to extract them so anyone could check for themselves.
The information is published annually and the data for 2014 have just been published.
So, has the decline of the previous 17 years been halted?
I've updated my charts to include the new data from 2014.



The complete data for these charts are (all these figures can be verified from the original HSCIC data):
|
Year |
Prescription Items |
Net Ingredient Cost |
Cost/item |
|
1995 |
164,207 |
£816,798 |
£4.97 |
|
1996 |
172,013 |
£914,983 |
£5.32 |
|
1997 |
162,421 |
£937,311 |
£5.77 |
|
1998 |
157,063 |
£927,633 |
£5.91 |
|
1999 |
147,769 |
£888,274 |
£6.01 |
|
2000 |
134,164 |
£831,130 |
£6.19 |
|
2001 |
127,333 |
£807,125 |
£6.34 |
|
2002 |
117,989 |
£778,749 |
£6.60 |
|
2003 |
103,940 |
£714,938 |
£6.88 |
|
2004 |
94,501 |
£661,469 |
£7.00 |
|
2005 |
82,960 |
£593,316 |
£7.15 |
|
2006 |
62,679 |
£442,769 |
£7.06 |
|
2007 |
49,316 |
£321,418 |
£6.52 |
|
2008 |
26,337 |
£152,300 |
£5.78 |
|
2009 |
19,005 |
£100,486 |
£5.29 |
|
2010 |
16,359 |
£121,449 |
£7.42 |
|
2011 |
15,501 |
£130,601 |
£8.43 |
|
2012 |
15,262 |
£143,068 |
£9.37 |
|
2013 |
13,001 |
£137,298 |
£10.56 |
|
2014 |
10,238 |
£110,438 |
£10.79 |
So, what does this show?
In 2014, the number of prescriptions for homeopathy fell for the eighteenth consecutive year, this time by over 21% — the fourth largest percentage fall since 1995 — continuing the downward spiral.
The number of prescriptions are now just 6% of what they were at their peak in 1996 — a fall of over 94%.
The total cost of these prescriptions also fell by 20% to a new low.
The cost per prescription has risen again, this time by 2%, compared to an increase of 13% in 2012/2013.
It's clear that doctors are writing fewer homeopathy prescriptions, but because these figures are for England only, the decisions by NHS Tayside and NHS Lothian in the past few years (and the recent decision by NHS Lanarkshire) to cease funding homeopathy referrals cannot explain this continued decline.
At the start of World Homeopathy Awareness Week, perhaps doctors are now more aware than ever that prescribing 'medicines' for which there is no good evidence can no longer be justified either on medical1 or ethical2 grounds.
References
1 “NHMRC Statement on Homeopathy and NHMRC Information Paper - Evidence on the Effectiveness of Homeopathy for Treating Health Conditions | National Health and Medical Research Council.” Accessed March 11, 2015. http://www.nhmrc.gov.au/guidelines-publications/cam02.
2 Shaw, David M. “Homeopathy Is Where the Harm Is: Five Unethical Effects of Funding Unscientific ‘remedies.’” Journal of Medical Ethics 36, no. 3 (March 1, 2010): 130–31. doi:10.1136/jme.2009.034959.
10 April 2015
Latest news
- "Undisputable evidence of scientific misconduct" by homeopaths
- Yet another bad year for homeopathy
- Nelsons Homeopathic Pharmacy #3
- Nelsons Homeopathic Pharmacy #2
- The Society of Homeopaths: failing to make the case for homeopathy
- The end of homeopathy on the NHS in Bristol?
- NHS Homeopathy: 20 years of decline
- The different faces of the Society of Homeopaths
- The growing pains of osteopaths
- Diluting misleading claims - ASA update
Most read
- Finding deleted and changed webpages
- About The Nightingale Collaboration
- How to find out who owns a website
- Advertising Standards Authority
- Rubbing salts into the wounds of homeopathy
- How to submit a complaint to the ASA
- The decline of homeopathy on the NHS
- Landmark decisions for homeopaths
- NHS Lanarkshire to end referrals to Glasgow Homeopathic Hospital
- Making a complaint



